Vaccination against HPV
Primary prevention of HPV
Several measures can help guard against HPV infection. However, they’re not all equally effective.
- Delaying the age of your first sexual encounter can help, but does not change the fact that your first partner might have had partners before you.
- Condoms can be very useful for preventing the spread of HPV, but do not cover all the surfaces that can be infected. Keep in mind that condoms are highly effective in preventing the spread of other STIs, as well as unwanted pregnancy!
- Reducing the number of your sexual partners can also help, but does not protect you against infected individuals with whom any partner might have had prior sexual contact.
- Quitting smoking does not protect against HPV. However, if you’re infected, your immune system fights the virus more effectively if you don’t smoke.
- The most effective way by far to protect yourself against HPV is to get vaccinated!
What does NOT protect against HPV?
- STI screening tests
- Many people are unaware that when you’re screened for an STI, the test does not include HPV. Many believe that the screening test includes the full range of STIs, but this is not the case.
- The Pap test, to screen for lesions that could lead to cervical cancer, is not effective in preventing the spread of HPV.
- The HPV screening test does not detect the low-risk HPV types associated with condylomas.
- Screening tests for high-risk HPV detect only those types that are in a replication phase. These tests are of little use in preventing the spread of HPV.
- Treating the lesions caused by HPV is not proven to reduce the risk of spreading HPV.
- Follow-up tests after treatment cannot determine whether you’re at risk of spreading HPV.
Vaccination against HPV
- Two vaccines are available, derived from the same principles of immunology.
- The vaccination works by injecting HPV protein into the body, which enables the formation of the external capsule of the HPV.
- • The vaccines contain no living virus or genetic material. It’s therefore impossible for them to transmit HPV or any related infection.
- • Both of these vaccines protect against HPV types 16 and 18.
- • Cervarix, a bivalent vaccine, protects against HPV types 16 and 18. Gardasil, a quadrivalent vaccine, also protects against these types, in addition to HPV 6 and 11.
Indications for HPV vaccines approved in Canada
Health Canada is responsible for establishing the rules that companies must follow in advertising their vaccines. Below, we provide the indications for each of the two HPV vaccines available in Canada.
1) Cervarix™
CERVARIX™ is a vaccine indicated in females from 10 to 25 years of age for the prevention of cervical cancer (squamous cell cancer and adenocarcinoma) by protecting against the following precancerous or dysplastic lesions caused by oncogenic Human Papillomavirus (HPV), types 16 and 18:
- Cervical intraepithelial neoplasia (CIN) grade 2 and grade 3
- Cervical adenocarcinoma in situ (AIS)
- Cervical intraepithelial neoplasia (CIN) grade 1
The effectiveness of Cervarix has not been studied in men. In women, this vaccine is effective only against cervical lesions.
2) Gardasil®
GARDASIL® is a vaccine indicated in girls and women 9- through 45 years of age for the prevention of infection caused by the Human Papillomavirus (HPV) types 6, 11, 16, and 18 and the following diseases associated with the HPV types included in the vaccine:
- Cervical, vulvar, and vaginal cancer caused by HPV types 16 and 18
- Genital warts (condyloma acuminata) caused by HPV types 6 and 11
- And the following precancerous or dysplastic lesions caused by HPV types 6, 11, 16, and 18:
- Cervical adenocarcinoma in situ (AIS)
- Cervical intraepithelial neoplasia (CIN) grade 2 and grade 3
- Vulvar intraepithelial neoplasia (VIN) grade 2 and grade 3
- Vaginal intraepithelial neoplasia (VaIN) grade 2 and grade 3
- Cervical intraepithelial neoplasia (CIN) grade 1
GARDASIL® is indicated in girls and women 9- through 26 years of age for the prevention of:
- Anal cancer caused by HPV types 16 and 18
- Anal intraepithelial neoplasia (AIN) grades 1, 2, and 3 caused by HPV types 6, 11, 16, and 18
GARDASIL® is indicated in boys and men 9 through 26 years of age for the prevention of infection caused by HPV types 6, 11, 16, and 18 and the following diseases associated with the HPV types included in the vaccine:
- Anal cancer caused by HPV types16 and 18
- Genital warts (condyloma acuminata) caused by HPV types 6 and 11
- Anal intraepithelial neoplasia (AIN) grades 1, 2, and 3 caused by HPV types 6, 11, 16, and 18
Dosage
Both HPV vaccines are usually administered in three doses over six months. You should not receive all three doses in less than a six-month period. However, if you miss a dose, you can receive it later, at your earliest convenience, without needing to restart the regimen.
Effectiveness and tolerance
- Both vaccines are highly effective in preventing new infections, as well as eliminating persistent infections and the lesions mentioned above.
- Most people have no reaction to either vaccine, however, Cervarix tends to cause more local reactions in the area where injected. Among women, Gardasil has shown to be effective in preventing more than 95% of HPV-related genital infections.
- Allergic reactions to either vaccine are rare.
- The most common side effect is soreness at the injection site.
- The vaccination schedule includes three doses.
- Females under 18 who are immunocompromised should receive a fourth dose of Gardasil under the free government program.
- Women should postpone the vaccination while pregnant or breastfeeding.
Quebec’s HPV vaccination program
- Since autumn 2008, the Gardasil vaccine had been administered to girls in the fourth grade of elementary school and in the third grade of secondary school.
- Vaccination is free of charge for girls under 18, as well as immunocompromised women up to age 26.
- Vaccination is free of charge for immunocompromised boys and men from 9 to 26.
The administration procedure is determined by your regional health board.
 Condyloma
Condyloma
 PDF
PDF




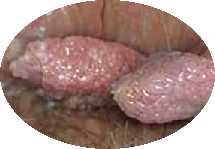 Condyloma
Condyloma
